To address the BCG shortage, ImmunityBio highlighted its recombinant BCG program, with 580 patients enrolled across about 100 clinical sites.
- ImmunityBio highlighted a “terrible” shortage of bladder-cancer therapy BCG with Merck as the lone global supplier.
- Anktiva Saudi launch preparations are underway, with product shipments ready following two approvals from the Saudi FDA in January.
- Net loss for FY25 narrowed to $351.4 million from $413.6 million in the previous year.
Shares of ImmunityBio, Inc. (IBRX) fell more than 5% in extended trading on Tuesday after the drugmaker warned of a “terrible” shortage of a standard therapy for early-stage bladder cancer, noting that Merck & Co., Inc. remains the sole supplier.
The company also said it is preparing shipments of its immunotherapy Anktiva to Saudi Arabia amid rising geopolitical tensions in the region following recent U.S.-Israel military action on Iran.
IBRX stock snapped three straight sessions of gains on Tuesday, declining over 4% to end at $10.
Anktiva Saudi Launch Preparations
During its earnings call, ImmunityBio said product shipments for the Saudi launch are ready to commence following two approvals from the Saudi Food and Drug Authority in January. The company said it will work closely with local authorities to manage imports as it prepares to launch the therapy in the country despite “escalating circumstances” in the Middle East.
“We recognize the ongoing global challenges, and especially those affecting the Middle East today, but cancer never pauses, and neither does ImmunityBio,” founder and executive chairman Patrick Soon-Shiong said.
ImmunityBio was granted two approvals in January, covering both bladder cancer and metastatic non-small cell lung cancer. The lung cancer authorization allows Anktiva to be used in combination with checkpoint inhibitors for metastatic disease.
The company also established a Saudi unit and partnered with regional distributors Biopharma and Cigalah to expand access across the Middle East and North Africa, which Soon-Shiong said represents “a significant and under-penetrated market for advanced immunotherapies.”
IBRX FY25 Review
On Tuesday, ImmunityBio reported a full-year net loss of $351.4 million, narrowing from $413.6 million in 2024. The company posted net product revenue of $113 million for 2025, up 700% from the previous year, following the commercial rollout of Anktiva. The company attributed the growth to increasing clinical adoption rather than price increases, noting that unit sales volume rose about 750% during the year.
Net product revenue rose to $38.3 million in the fourth quarter, up 20% from the previous quarter. The company said each quarter of 2025 delivered higher revenue than the prior period. ImmunityBio ended the year with $242.8 million in cash, cash equivalents, and marketable securities.
ImmunityBio’s Response To BCG Shortage
The company highlighted the ongoing shortage of Bacillus Calmette-Guerin (BCG), the decades-old therapy widely used to treat non-muscle invasive bladder cancer. It noted that Merck is currently the only supplier of BCG globally.
ImmunityBio pointed to its recombinant BCG program as a potential solution. The company said the U.S. Food and Drug Administration (FDA) authorized an expanded access program in February 2025 to help address the shortage of Tumor Immunotherapy with BCG.
According to the drugmaker, the first patient was treated with recombinant BCG in March 2025, and 580 patients had been enrolled in the program as of February. The program now includes 100 clinical sites across academic centers and community urology practices, with “several thousand” doses administered either as monotherapy or in combination with Anktiva.
ImmunityBio has requested a meeting with the FDA scheduled this month to discuss the next steps for recombinant BCG development.
ImmunityBio’s AI Push
The company also outlined its internal AI platform, “Ask IB,” which integrates large language models and automated systems across its research and manufacturing operations.
ImmunityBio said the platform is being used to support robotic manufacturing processes, scaling production of immune cell therapies while reducing contamination risk and enabling continuous production. It will also support predictive analytics across research and development and commercial operations.
How Did Stocktwits Users React?
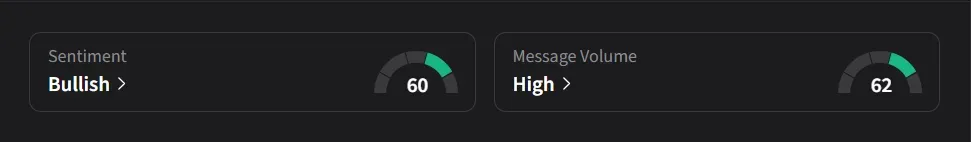
On Stocktwits, retail sentiment for IBRX was ‘bullish’ amid ‘high’ message volume.
One user said, “I would say if orders are ready to ship to Saudi Arabia and Germany, I see no reason for any dilution. The revenues will continue to scale.”
Another user said, “Earnings and earnings call were both GREAT. Ignore the crazy fluctuations which ALWAYS happen in biotech investing and we have a huge war going on in the Middle East effecting the mkts. All you have to do is HODL and get RICH!”
IBRX shares have climbed more than 400% year-to-date, far outperforming the Nasdaq’s 10% decline and the S&P 500’s 0.5% gain.
For updates and corrections, email newsroom[at]stocktwits[dot]com.<